The Science
Blood, DNA, and the gut microbiome are the three biological systems that shape your child's health before birth, through infancy, and across a lifetime.
Powered by Independente Research

Blood, DNA, and the gut microbiome are the three biological systems that shape your child's health before birth, through infancy, and across a lifetime.
Powered by Independente Research

A baby's early development is shaped entirely by the mother's biological environment. Three systems play a particularly important role - the gut microbiome, maternal blood biology, and the epigenetic signals written into a baby's DNA from the earliest weeks of pregnancy.
Modern research shows that the biological environment during pregnancy helps shape a newborn's immune system, metabolism, digestive health, cognitive development, and early resilience - with effects that extend across childhood and into adult life.
The composition of a mother's blood - her nutrient levels, hormones, inflammatory markers, and immune proteins - directly shapes the biological environment a baby grows within. From pre-conception through postnatal recovery, blood biomarkers provide a window into the systems that matter most. Deficiencies that go undetected can have lasting effects on pregnancy outcomes, baby development, and long-term wellbeing.
DNA is not destiny alone - it is a blueprint whose expression is continuously shaped by the biological environment. Epigenetics is the science of how nutrients, hormones, stress, and microbial signals during pregnancy chemically modify gene expression. The biological conditions present during pregnancy and early life have a lasting influence on how a child's genes are expressed for decades to come.
The gut microbiome - the ecosystem of trillions of microorganisms living in the digestive tract - is one of the most biologically influential systems in the human body. During pregnancy, the maternal microbiome shifts dramatically to support foetal development. At delivery, this ecosystem is transferred to the newborn, seeding the infant gut and programming immune, metabolic, and neurological development for years to come.
Blood, DNA, and gut don't just affect pregnancy - they are the biological architects of a child's immune system, brain, metabolism, and mental health across every stage of childhood and beyond.
A child's immune system is educated by the microbial environment, shaped by nutritional status, and programmed by epigenetic signals that begin in the womb. How robust, balanced, and tolerant it becomes is directly tied to the biology of early life.

Neurodevelopment begins in the first weeks of pregnancy. The gut-brain axis, maternal nutritional status, and epigenetic programming converge to shape cognitive development, language acquisition, attention, and sensory processing from the earliest moments.
The "developmental origins of health and disease" concept - backed by decades of research - shows that metabolic conditions including obesity, type 2 diabetes, and cardiovascular disease have their biological roots in the prenatal environment.

Anxiety, ADHD, depression, and behavioural difficulties have measurable biological underpinnings established in the first years of life, rooted in the gut, blood, and DNA.

The interplay of blood, DNA, and gut shapes four core areas of child development - and Kids BioCare measures all three drivers.
The immune system is educated by microbial exposure, regulated by nutritional biomarkers, and epigenetically calibrated in utero. Imbalances here underlie allergy, eczema, asthma, and autoimmune conditions.
From neural tube formation to neurotransmitter production - brain development is shaped by blood nutrients, gut microbes, and epigenetic programming from the earliest weeks.
How a child's body manages energy, stores fat, and responds to insulin is set in the first thousand days - through epigenetic imprinting, microbial energy harvest, and maternal hormonal signalling.
Anxiety, mood, attention, and resilience emerge from biological systems established in early life - with the gut-brain axis, stress-response epigenetics, and nutritional co-factors as the primary drivers.
The biological connections between maternal health, gut, blood, DNA, and child outcomes are not theoretical. Here is a selection of the peer-reviewed research that underpins the Kids BioCare approach.
A cohort of 1,288 mother-infant pairs found that infant gut microbiome composition at 1 week was directly predictable from the maternal microbiome. Vaginally-born infants showed significantly higher Bifidobacterium colonisation and superior IgA mucosal immunity at 6 months.
Double-blind RCT supplementing pregnant women with DHA from week 18 through 3 months postpartum. Children of DHA-supplemented mothers showed significantly higher mental processing scores at age 4 and better problem-solving at 6 months.
The Avon Longitudinal Study (3,500+ children) found that children exposed to high prenatal maternal stress showed measurable methylation of the glucocorticoid receptor gene - correlated with a 60% increased rate of ADHD-type symptoms and anxiety disorders at ages 7 and 10.
A Canadian cohort identified four bacterial genera whose combined abundance at 3 months was inversely correlated with asthma risk by age 7. Children deficient in these species showed 3x higher rates of asthma and atopic sensitisation in school years - confirmed in animal models.
Prospective birth cohort of 1,200 mother-child pairs. Children born to Vitamin D-insufficient mothers had a 40% higher rate of asthma by age 5 and significantly lower regulatory T-cell function - independent of breastfeeding, birth mode, and allergen exposure.
Two landmark studies established a direct causal gut-brain link. Gut enterochromaffin cells produce 95% of total body serotonin in a microbiome-dependent manner. L. rhamnosus supplementation from birth reduced anxiety behaviour and modified GABA receptor expression in the brain.
Over 30 years of evidence established that adult cardiovascular disease, type 2 diabetes, and obesity have origins in the prenatal nutritional environment. Children born small-for-gestational-age show epigenetic upregulation of fat-storage genes - creating a metabolic phenotype harmful across a lifetime.
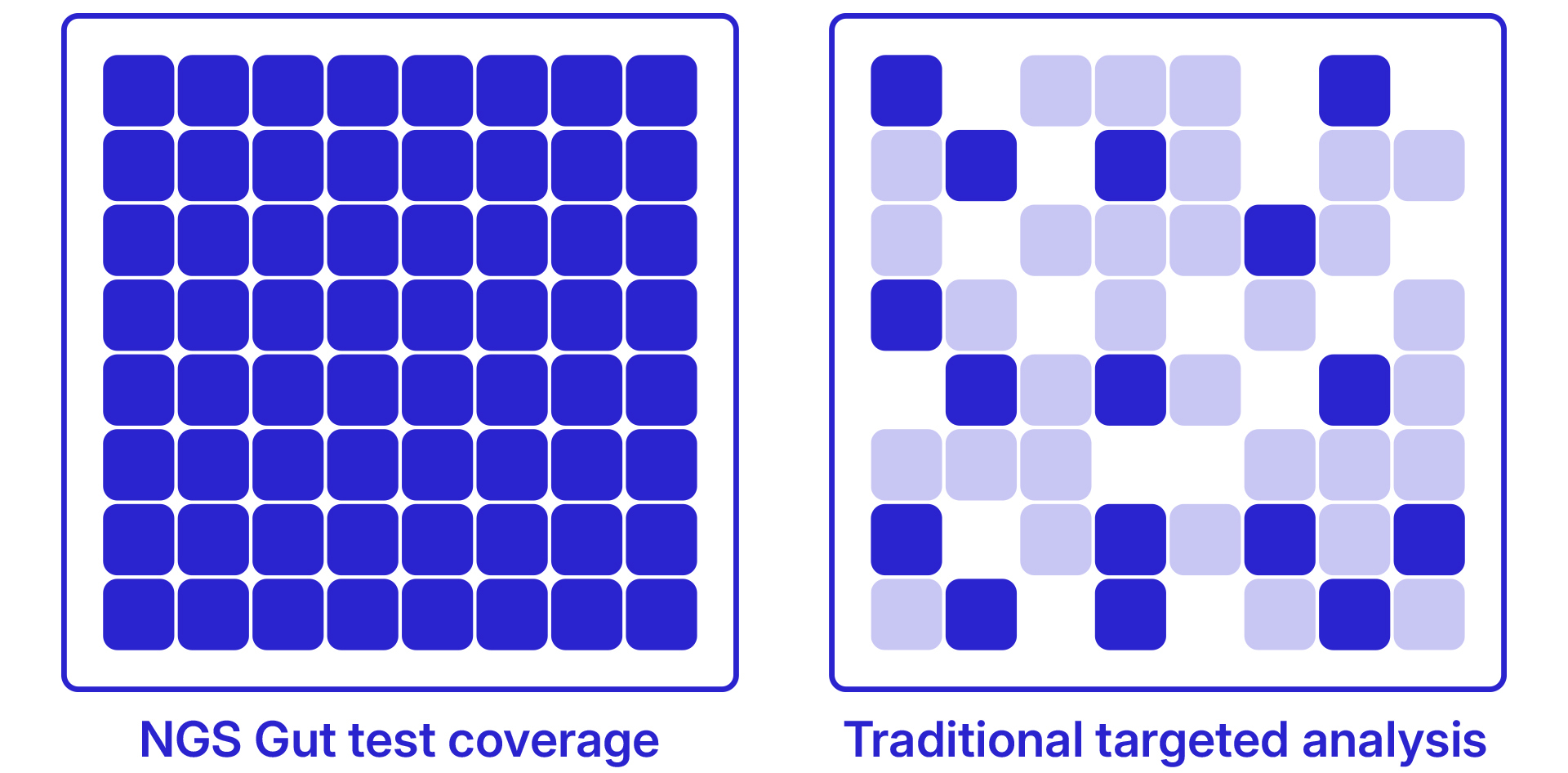
Study of 600 children aged 2-6 demonstrated that Akkermansia muciniphila abundance - measurable only through NGS sequencing - was inversely correlated with BMI, fasting insulin, and inflammatory markers. Children in the lowest quartile were 2.4x more likely to show early metabolic syndrome markers.
Longitudinal study following 191 children from infancy to age 19. Iron deficiency in the first year - even without overt anaemia - caused lasting deficits in cognitive function, academic achievement, and emotional regulation. Brain imaging revealed reduced myelination in the hippocampus. Effects persisted even after iron levels were later normalised.
Kids BioCare integrates advanced NGS microbiome sequencing, biomarker blood testing, and EatIQ nutrition intelligence - all delivered through BioHealthcare Hub with guidance from specialist doctors.
Kids BioCare gives families the biological insights to understand that process - from pre-pregnancy through early childhood. Because the choices made during these early years may shape your child's health for a lifetime.